European clinical AI companies are generating real-world results far from the headlines, and the clearest current evidence is a collaboration anchored in Germany and Switzerland. The three-year AI collaboration MoU signed on 20/04/2025 between Sidra Medicine, Vitafluence.ai GmbH (registered in Germany) and Swiss venture studio EmpathicAI.Life is now in active clinical operation. As of early 2026, that agreement is no longer a letter of intent; it is the structural backbone of a functioning paediatric AI programme. European AI observers and hospital procurement teams should pay close attention to what this partnership is demonstrating about deployment methodology.
Vitafluence anchors the precision medicine push
The MoU covers predictive modelling, AI-assisted diagnostics, and tailored treatment pathways. For clinicians, the practical result is access to tools that support clinical decisions, personalise patient care, and flag cases earlier. For a hospital whose mission centres on paediatrics, rare conditions, and maternal health, that is a substantive capability upgrade, not an incremental one.
Dr. Peter O. Owotoki, Co-Founder of Vitafluence.ai, has described the collaboration as an opportunity to contribute to ambitious medical and digital health advancement, with a focus on delivering impactful outcomes for women and children. That framing is consistent with the direction European clinical AI developers have been pushed toward by both the EU AI Act and national health-technology assessment bodies: demonstrate clinical impact in defined populations, not broad-spectrum capability claims.
The precision medicine framing is worth unpacking for a European audience. The EU's Personalised Medicine 2030 Action Plan, backed by the European Commission, and the Swiss Federal Office of Public Health's ongoing digital health strategy both position precision medicine as the organising principle of next-generation healthcare. Vitafluence.ai building validated AI tools in this domain positions it well for European regulatory and procurement conversations, particularly under the EU AI Act's high-risk medical device provisions.
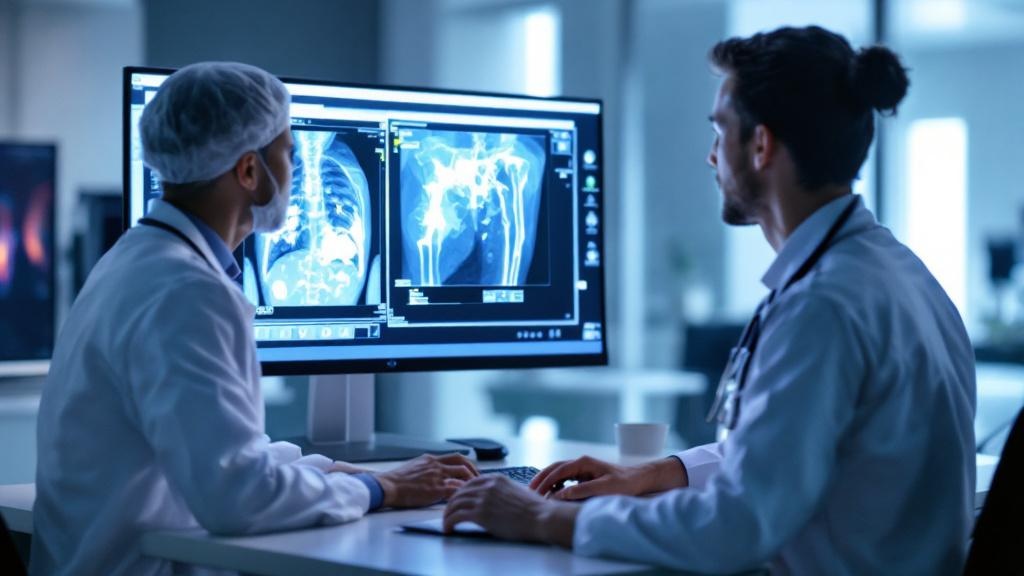
Paediatric emergency AI is already live
The most operationally significant detail is that an AI tool for detecting fractures and dislocations in paediatric imaging is deployed and running inside an Emergency Department and Diagnostic Imaging Unit. This is not a pilot. It is in production, targeting a high-volume, high-stakes workflow where missed diagnoses carry lasting clinical consequences.
Paediatric imaging is one of the most technically demanding domains for diagnostic AI. Paediatric anatomy changes rapidly with age, bone density differs substantially from adult norms, and scan protocols are often constrained by the need to minimise radiation exposure. A clinically validated fracture-detection system for paediatrics represents genuine engineering and regulatory rigour, not a straightforward adaptation of an adult imaging model.
For European hospital administrators, this is instructive. The UK's NHS AI Lab, part of NHS England, has repeatedly emphasised in its evaluation frameworks that the critical question for any clinical AI tool is not whether it works in a controlled study but whether it performs reliably inside a live clinical workflow under real operating pressures. Vitafluence.ai's paediatric fracture detection deployment clears that bar. Professor Mihaela van der Schaar, Director of the Cambridge Centre for AI in Medicine at the University of Cambridge, has similarly argued in published work that AI tools validated only in retrospective datasets remain a category apart from those integrated into prospective clinical decision-making. This deployment sits in the latter category.
Three reasons this matters for European healthcare AI procurement:
- It demonstrates a European company operating inside a regulated clinical environment at production scale.
- It targets a workflow, paediatric emergency imaging, where European hospitals carry comparable diagnostic pressures.
- It gives Vitafluence.ai directly transferable evidence for CE marking and UKCA submissions under the Medical Device Regulation framework.
Research integration: the annual forum model
The partnership is not limited to clinical deployment. An Annual Research Forum in January 2026 included a dedicated workshop on AI in healthcare, covering AI-assisted scientific writing and peer review. For a research hospital, embedding AI into both care delivery and research methodology simultaneously is the 2026 baseline expectation, not an ambitious target.
This dual-track approach, clinical and academic, is directly relevant to European academic medical centres. Institutions such as the Charité in Berlin, the University Hospital Zurich, and the Karolinska in Stockholm are all navigating the same question: how do you integrate AI tools into clinical routine without fracturing your research infrastructure? The Sidra-Vitafluence model suggests one answer: build the research forum into the deployment programme from the outset, so that clinical use generates the academic output, rather than treating the two as separate workstreams.
Key milestones at a glance
- 20/04/2025: Three-year MoU signed between Sidra Medicine, Vitafluence.ai GmbH, and EmpathicAI.Life covering predictive modelling, diagnostics, and tailored treatment pathways.
- In production (2025-2026): Paediatric fracture and dislocation detection AI deployed in Emergency Department and Diagnostic Imaging Unit.
- 27-28/01/2026: Annual Research Forum featuring dedicated AI in Healthcare workshop.
- Ongoing: Precision medicine research aligned with national health strategy targets.
What European hospital AI teams should take from this
The methodology here is replicable and instructive for European health systems. Start with a focused international technical partnership. Target a single high-value clinical workflow. Build internal research capacity through a recurring annual forum. Embed the work in a national or institutional strategy so that budget and political support are protected across procurement cycles.
That sequence avoids the most common failure mode in European hospital AI deployments: attempting a system-wide rollout before any single workflow is validated, then losing momentum when the first use case underperforms. The NHS has seen this pattern repeatedly. So have several German Unikliniken that ran broad AI pilots in 2022 and 2023 without a focused workflow anchor.
The deeper question for European AI developers is whether Vitafluence.ai's production deployment can be used as a reference installation for procurement conversations in Germany, Switzerland, and the UK. Given the EU AI Act's requirements for post-market monitoring of high-risk AI systems in healthcare, having a live deployment with documented clinical performance is not just a commercial advantage; it is increasingly a regulatory prerequisite. EmpathicAI.Life's involvement as a venture studio also points toward a model where early-stage AI companies can access clinical environments through structured MoU arrangements rather than waiting for full commercial contracts, a pathway that European hospital innovation offices should be actively considering.
The detail that separates this programme from noisier announcements elsewhere is simple: the fracture detection tool is running. In 2026, that remains the meaningful distinction in clinical AI.



Comments
Sign in to join the conversation. Be civil, be specific, link your sources.